(1-Imidazolyl)acetonitrile CAS 98873-55-3 Luliconazole Intermediate Purity ≥99.0% (HPLC)
Supply Luliconazole Related Intermediates
Luliconazole CAS 187164-19-8
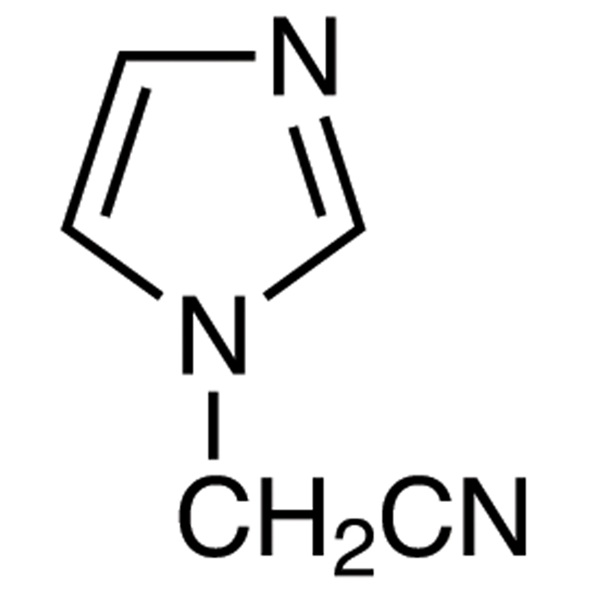
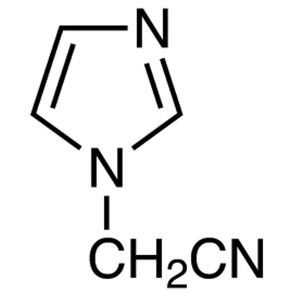
(1-Imidazolyl)acetonitrile CAS 98873-55-3
(S)-2,4-Dichloro-α-(Chloromethyl)benzyl Alcohol CAS 126534-31-4
| Chemical Name | (1-Imidazolyl)acetonitrile |
| Synonyms | 1-(Cyanomethyl)imidazole; Imidazol-1-YL-Acetonitrile; 1-Imidazolylacetonitrile; 1H-Imidazole-1-Acetonitrile |
| CAS Number | 98873-55-3 |
| Stock Status | In Stock, Production Scale Up to Tons |
| Molecular Formula | C5H5N3 |
| Molecular Weight | 107.12 |
| Melting Point | 54.0 to 58.0℃ |
| Density | 1.11±0.10 g/cm3 |
| COA & MSDS | Available |
| Place of Origin | Shanghai, China |
| Product Categories | Pharmaceutical Intermediates |
| Brand | Ruifu Chemical |
| Item | Specifications |
| Appearance | White to Off-White Powder |
| ldentification By HPLC | The principle peak retention time of the sample should match with that of the standard |
| Purity / Analysis Method | ≥99.0% (HPLC) |
| Loss on Drying | ≤0.50% |
| Related Substances | By HPLC |
| Single Impurity | ≤0.50% |
| Total Impurities | ≤1.00% |
| Infrared Spectrum | Conforms to Structure |
| NMR Spectrum | Conforms to Structure |
| Test Standard | Enterprise Standard |
| Usage | Intermediate of Luliconazole (CAS: 187164-19-8) |
Package: Bottle, Aluminium foil bag, 25kg/Cardboard Drum, or according to customer's requirement.
Storage Condition: Store in sealed containers at cool and dry place; Protect from light and moisture.

UN IDs UN 3439 6.1/PG III
Hazard Class 6.1
Packing Group III
(1-Imidazolyl)acetonitrile (CAS: 98873-55-3) is an intermediate of Luliconazole (CAS: 187164-19-8). (1-Imidazolyl)acetonitrile is also a reactant that has been used in the synthesis of Lanoconazole, an antifungal. Luliconazole is a novel topical antifungal imidazole, and is a kind of analogue of Lanoconazole. It can interfere with the fungal cell wall synthesis and fungal growth by decreasing levels of ergosterol via inhibiting lanosterol demethylase activity. Luliconazole was originally developed by the Japanese pesticide Corporation (NihonNohyaku Co., Ltd.). In November 2013, the FDA has approved a 1% Luliconazole cream for entering into market for topical treatment of interdigital athlete's foot, jock itch and ringworm.